Certification Methods
It is important that our customers know that the value given on the certificate attached to the cylinder is accurate.
The UK gases industry uses two main ways of assigning a certificated value to a gas mixture.
The first is by careful measurement of the weight of the gases being filled into the cylinder. The second is to analyse the contents by withdrawing product from the cylinder for post-fill analysis.
Certification by weight
Certification by weight (often called gravimetric certification), claims high filling tolerance and high certification accuracy.
The modern mass comparators used by BOC have a resolution of 0.002g in 30000g. For this reason gas mixtures filled in this way can be said to offer an accuracy of ± 1%.
With gravimetrically prepared mixtures this filling tolerance is often quoted as the certification accuracy.
Certification by analysis
Certification by analysis involves analysing the gas from a cylinder and comparing the result of that analysis to a known gas standard analysed on the same instrument. The degree of accuracy of the analysis determines the grade of the product sold.
BOC will choose the best combination of filling and analytical methods to meet customer specifications.
The advantage of analysis is that the gas mixture which the customer will be using will have been directly sampled and tested.
Certification grades
Accredited Reference Materials manufactured to ISO 17034 and certified to ISO/IEC 17025
Ethanol / air and MOT mixtures certified to +/- 1% relative, and a range of binary, natural gas and gravimetric mixtures with individually calculated measurement uncertainties are available as accredited calibration mixtures.
 0408 |
 4183 |
| Alpha Standard | Certified value within +/- 1% of the actual concentration value e.g. for an analytical result of 10% CO2 in N2 the CO2 will be between 9.9% and 10.1% |
| Beta Standard | Certified value within +/- 2% of the actual concentration value e.g. for an analytical result of 10% CO2 in N2 the CO2 will be between 9.8% and 10.2% |
| Certified Mixture | Individual cylinder analysis certified to be within +/- 2% to +/- 5% of the actual concentration value |
| Uncertified Mixture | Individual or batch analysis may be performed but no analysed value is quoted |
Traceability
Traceability is an unbroken chain of comparison leading to an SI Unit of measurement. In the case of gases this the kilogram.
Metrological traceability is “The property of the result of a measurement or the value of a measurement standard that relates it to a stated reference, usually a national or international standard, through an unbroken chain of comparisons with stated uncertainties”
Why is traceability necessary?
Traceability is a tool used to provide accuracy and comparability of measurement results at both national and international levels.
Traceable gas mixtures
A traceable gas reference mixture is one whose concentration has been determined through a chain of direct comparisons to national measurement standards for the particular type of mixture.
By having documented links to a common, highly accurate point of reference, all traceable mixtures have both demonstrable accuracy and links to each other.
As the national standards are regularly compared with those of other countries, this network of traceable mixtures extends internationally, enabling measurement results to be accepted internationally
Benefits of traceable calibrations
Traceable calibrations automatically provide the means to obtain:
Consistent data over time, even when one mixture is replaced by another
Comparability of data obtained from similar measurements elsewhere, even internationally; and
Known accuracy in SI Units (molar parts per million)
Hierarchy of Gas Calibration Standards
- SI Units is the International System of Units
- National Reference Laboratory & Measurement Standards are provided by organisations such as NIST, NPL and NMI (see below)
- QA/QC Calibration Laboratories are typically those with ISO 17025 and ISO 17034 accreditation. This includes BOC
- Field measurements are carried out by customers of BOC using, for example, accredited gas calibration mixtures
There are two principal ways to obtain traceability when filling a calibration gas mixture:
- by weight
- by analytical comparison to a known standard

Traceability by analysis
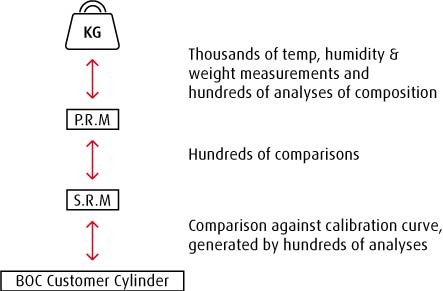
Traceability by analysis is essentially derived via a careful series of comparisons between gas sampled from the customer's mixture cylinder with gas sampled from a reference cylinder prepared by a national standards body, such as the National Physical Laboratory.
Many countries have national standards institutes. The principal ones concerning gas analysis are:
- the National Physical Laboratory (NPL), in the UK
- the National Institute of Science and Technology (NIST), in the US
- the Netherlands Metrology Institute (VSL)
The NPL, NIST, and VSL all prepare primary reference materials (PRMs). This is done initially by weight, thus forming the link with the SI unit of mass under tightly controlled and measured conditions of temperature, humidity and pressure.
Each PRM is subsequently analysed and compared with other PRMs and standards under controlled conditions.
When all of this data is collated the PRM can be said to be a particular value with a low level of measurement uncertainty.
Primary Reference Materials (PRMs) are maintained within the institution that made them and are used to produce Secondary Reference Materials (SRMs) which are sold to, amongst others, BOC.
These SRMs are produced under similar conditions as the PRMs. BOC uses the SRM to analyse its cylinders and assign certificate values to customer cylinders, thus forming the unbroken chain of comparison to the kilogram mass.
Our process for producing standards from SRMs is highly detailed in its assessment of the BOC cylinder. SRMs are used to plot correlation curves of the analyser response. Each curve is frequently updated and checked through comparison with other SRMs and internally prepared gravimetric standards. These standards are used to re-check the curve validity every time a customer's cylinder is analysed and certified.
By comparing multiple samples of gas from the customer's cylinder with this suite of measurements we are assured a very high level of confidence in the result we assign to the calibration mixtures we sell and can prove an unbroken chain of comparison or traceability.
Traceability by weight (gravimetric)
Traceability by weight involves comparing the weight of gas in the cylinder with a mass that has itself been compared with one that has been compared to the standard SI mass.
Provided that the manner in which these comparisons are made is documented and subject to audit this is an acceptable way to obtain traceability of the measurements made to produce the gravimetrically prepared gas mixture.



